Ferritinophagy: unveiling a Druggable Nexus in Quiescent Leukemic Stem Cells: A Leap Toward Curbing Acute Myeloid Leukemia
In the intricate realm of Acute Myeloid Leukemia (AML), researchers have long grappled with the enigmatic nature of Leukemic Stem Cells (LSCs) which, despite their scarce presence, cast a long and malignant shadow. Their ability to evade conventional therapies by entering a quiescent state has been a significant hurdle in achieving enduring remissions in AML. The recent exploration by Clement Larrue and colleagues, titled "Ferritinophagy is a Druggable Vulnerability of Quiescent Leukemic Stem Cells," published as a pre-print on ScieNFT heralds a promising stride toward unmasking the vulnerabilities of these quiescent LSCs, thereby nudging the field closer to efficacious therapeutic interventions.
Larrue et al., through a meticulous series of experiments, illuminate the understudied domain of quiescent cells in AML, especially accentuating their iron metabolism and autophagy-dependent survival mechanism. They used patient-derived xenograft (PDX) models and administered Bromodeoxyuridine (BrdU) for cellular proliferation marking over a week. They confirmed that BrdU incorporation was notably higher in the cycling AML cells than the quiescent ones. Yet, even quiescent cells displayed evidence of prior cell cycle activity. A deeper dive into the phenotypically defined quiescent CD34+CD38− cell subpopulation revealed it to be predominantly quiescent when compared to CD34+CD38+ cells in vivo (Figure 1G-H). In subsequent in vivo examinations, we separated AML cells based on their cycling status. Both types were then xenotransplanted into NSG mice while early post-transplantation phase didn't reveal notable differences, the quiescent fraction demonstrated a heightened engraftment capacity after a period of 8-12 weeks (Figure 1J). This suggests an enrichment of leukemia-initiating cells within the quiescent subset.
This finding highlights the unique presence of quiescent leukemic stem cells (LSCs) in PDX models.
The authors then employed a transcriptomic and single-cell analyses to discern unique features in sorted quiescent cells. The gene set enrichment pinpointed an enrichment in LSC-related genes within the quiescent group. They derived a novel quiescent AML up (Q_AML_UP) signature encompassing 37 genes which had prognostic value within the TCGA cohort (Figure 2 A-C).
Their subsequent analysis showed upregulation of the autophagy pathway in the quiescent cells (Figure 3A).
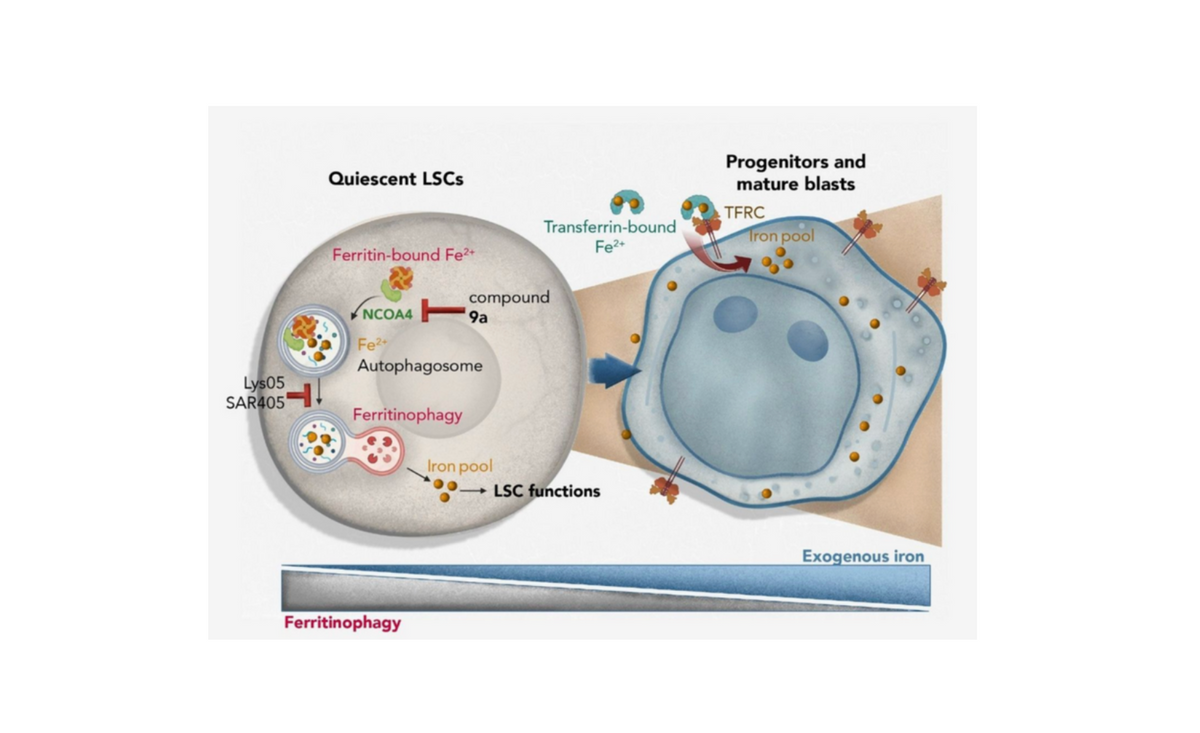
The role of autophagy in iron metabolism, especially in degrading iron-containing proteins, was explored in both dormant and active cell groups. Using specific inhibitors, a significant expanded iron pool was observed in active cells, and iron bioavailability was found to be decreased by autophagy suppression (See Figures 3D and 3E). Autophagy-dependent iron availability seemed vital for the dormant leukemic cell subset and is essential for the optimal functioning of progenitor and LSCs in AML.
The revelations regarding the interplay between ferritinophagy— the process of ferritin degradation to liberate iron—and the survival of quiescent LSCs, unveils a unique metabolic dependency that could be exploited for therapeutic advantage.
The narrative of the research unfolds elegantly as it delineates the role of Nuclear Receptor Coactivator 4 (NCOA4) in orchestrating ferritinophagy. The heightened expression of NCOA4 in quiescent cells, as opposed to their cycling counterparts, underscores a potential therapeutic target. The subsequent introduction and exploration of Compound 9a, which impedes NCOA4’s function, thereby disrupting ferritinophagy, is particularly riveting (Figure 6K-N).
The profound impact of this inhibition on the survival of quiescent LSCs, both in vitro and in vivo, is a testament to the potential of this therapeutic avenue. Moreover, the nuanced understanding provided by the study regarding the unique iron metabolism of quiescent LSCs adds a valuable layer to our existing knowledge. It's not just the unveiling of the NCOA4 as a target, but the meticulous delineation of the molecular machinations that sustain the quiescent LSCs, that makes this work a notable contribution to the field.
Ferritinophagy is a cellular process that involves the degradation of ferritin, which is the primary iron storage protein in cells. This process is crucial for the maintenance of iron homeostasis in the body. Ferritinophagy plays a role in various cellular processes and has been implicated in several diseases, including cancer. Here's a brief summary based on the insights from different sources:
Ferritinophagy might play a role in the maintenance and function of cancer stem cells. Although the exact relationship between ferritinophagy and CSCs is not entirely clear, it's believed that targeting ferritinophagy could provide a novel approach for cancer therapy.
Ferritinophagy has also been associated with ferroptosis, a type of programmed cell death dependent on iron. By degrading ferritin and releasing iron, ferritinophagy can contribute to the ferroptotic process, which has been proposed as a therapeutic strategy against cancer, including targeting CSCs that are known for their resistance to conventional therapies. The rigor and breadth of the investigations undertaken by Larrue et al., paired with the clarity of their insights, carve a clear path toward understanding and targeting the elusive quiescent LSCs in AML. The translational implications of their findings, especially the potential of NCOA4 inhibition as a therapeutic strategy, could indeed be a significant step forward in the relentless pursuit of durable remissions in AML.
By delving into the metabolic dependencies of these dormant yet menacing cells, Larrue and team have crafted a narrative that is as enlightening as it is promising for the future of AML treatment.
Press Release
Title: "Discovering Hidden Mechanisms in Leukemia Cells: A Step Towards Understanding Acute Myeloid Leukemia"
Researchers Clement Larrue, Jerome Tamburini, and their team have recently published a significant study on ScieNFT, a decentralized pre-print platform, revealing a hidden mechanism in a certain type of leukemia cells. The study dives into the world of Acute Myeloid Leukemia (AML), a severe blood cancer, to unravel how some of its cells manage to evade current treatments and continue to thrive.
The focus here is on a special group of leukemia cells known as quiescent Leukemic Stem Cells (LSCs). Unlike regular cells, these LSCs decide to take a break and go into a sort of hibernation, making them tough targets for existing treatments. This "sleep mode" allows them to escape the effects of chemotherapy, one of the main treatments for AML, and resurge later, making the disease return.
The researchers have discovered a particular mechanism, named ferritinophagy, that seems to play a crucial role in keeping these quiescent LSCs alive during their hibernation. Ferritinophagy is a process that helps manage iron levels within the cells by breaking down a protein called ferritin. This mechanism, along with a molecule named NCOA4, appears to be a key to the survival strategy of these elusive leukemia cells.
The exploration of ferritinophagy and its interaction with NCOA4 unveils a new layer of complexity in understanding AML. This discovery provides a glimpse into the resilience and survival strategies of leukemia cells, emphasizing the importance of continuous exploration in the fight against AML. Through studies like these, researchers hope to gather crucial insights that could inform future strategies to combat this challenging disease.
We are immensely honored at ScieNFT to host such a remarkable research paper. Indeed, by sharing their scientific findings in an open access format, these researchers are significantly advancing the cause of decentralized open science.